Thermodynamics
Define system and surrounding.
Substance which is taken under observation is called system , rest of the universe is called surrounding.
Explain the different types of system with example.
There are three types of the system.
(i) Open system.
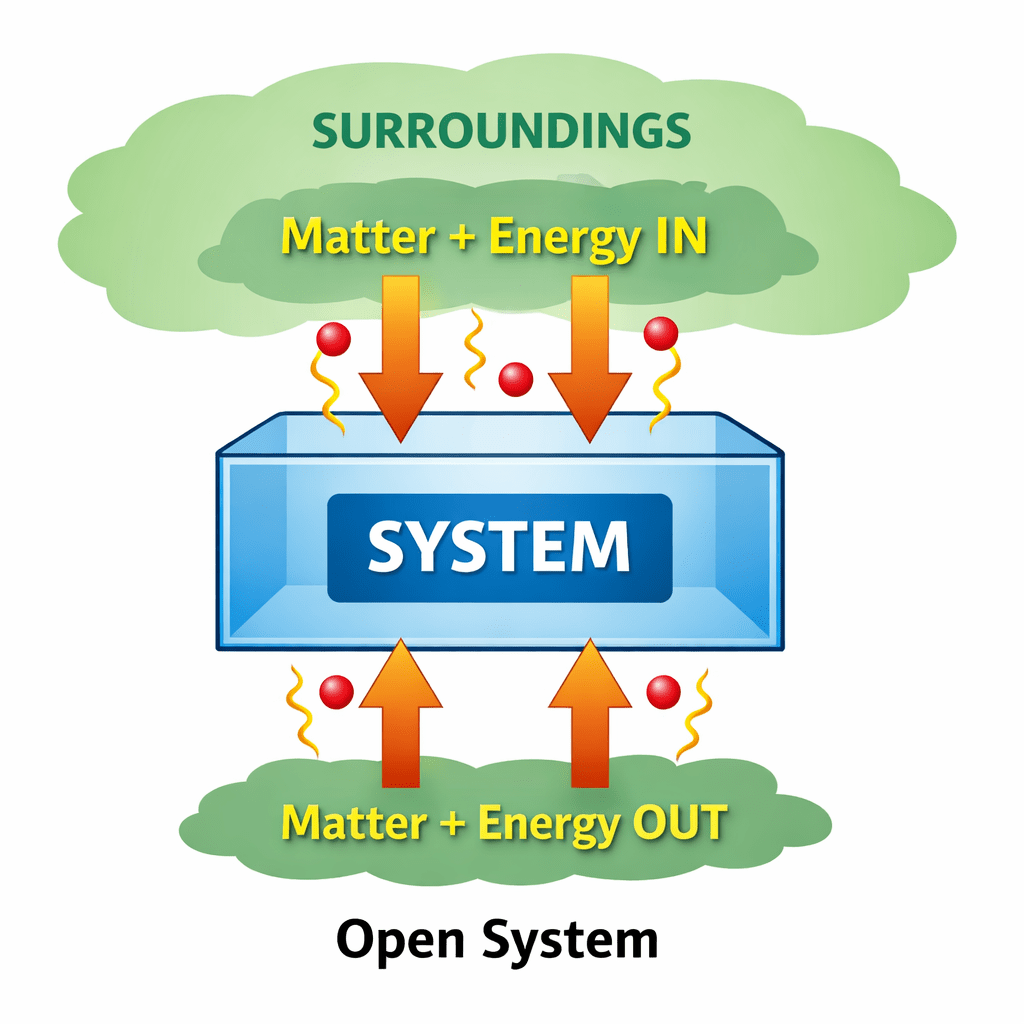
The type of system in which both matter and energy is exchanged between system and surroundings.
Example: Substance in open container.
(ii) Closed system.
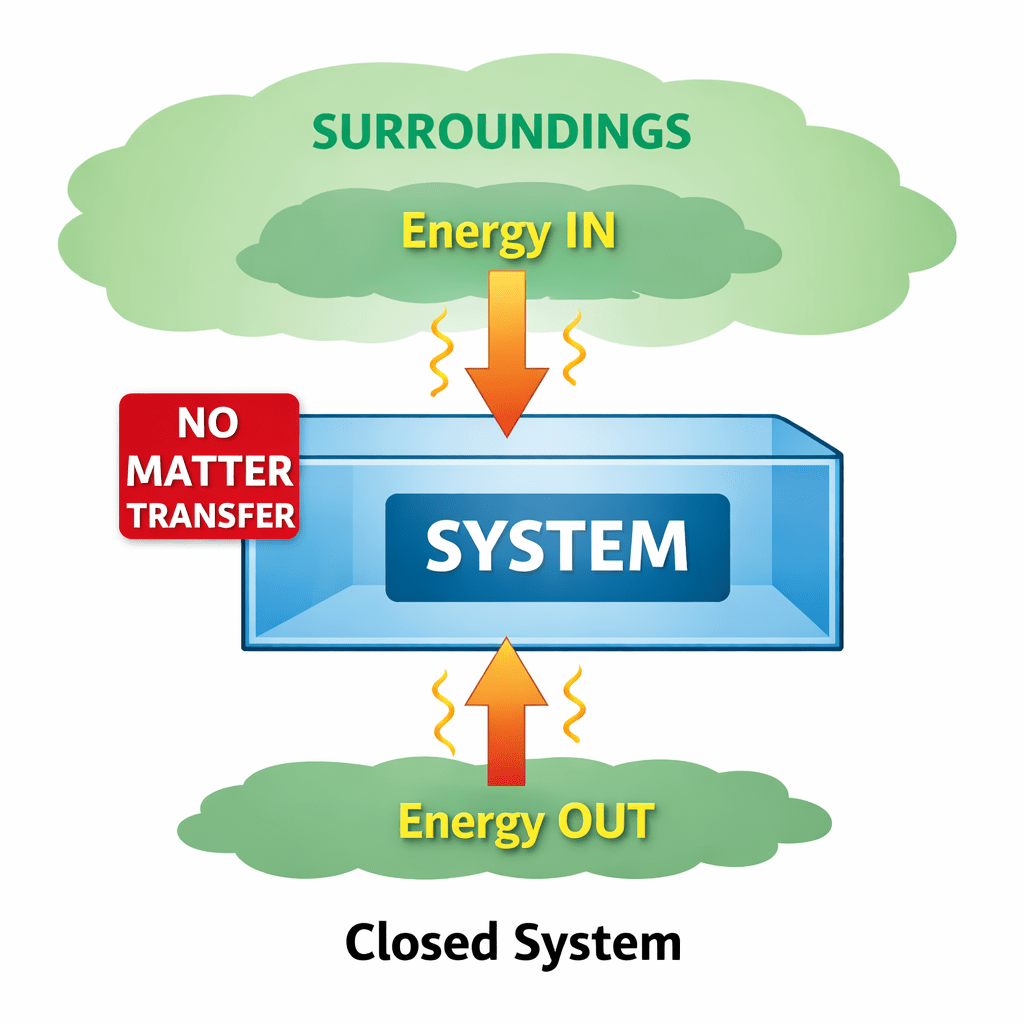
Example: Substance in close container having thermoconducting wall.
The type of system in which only energy is exchanged between system and surroundings.
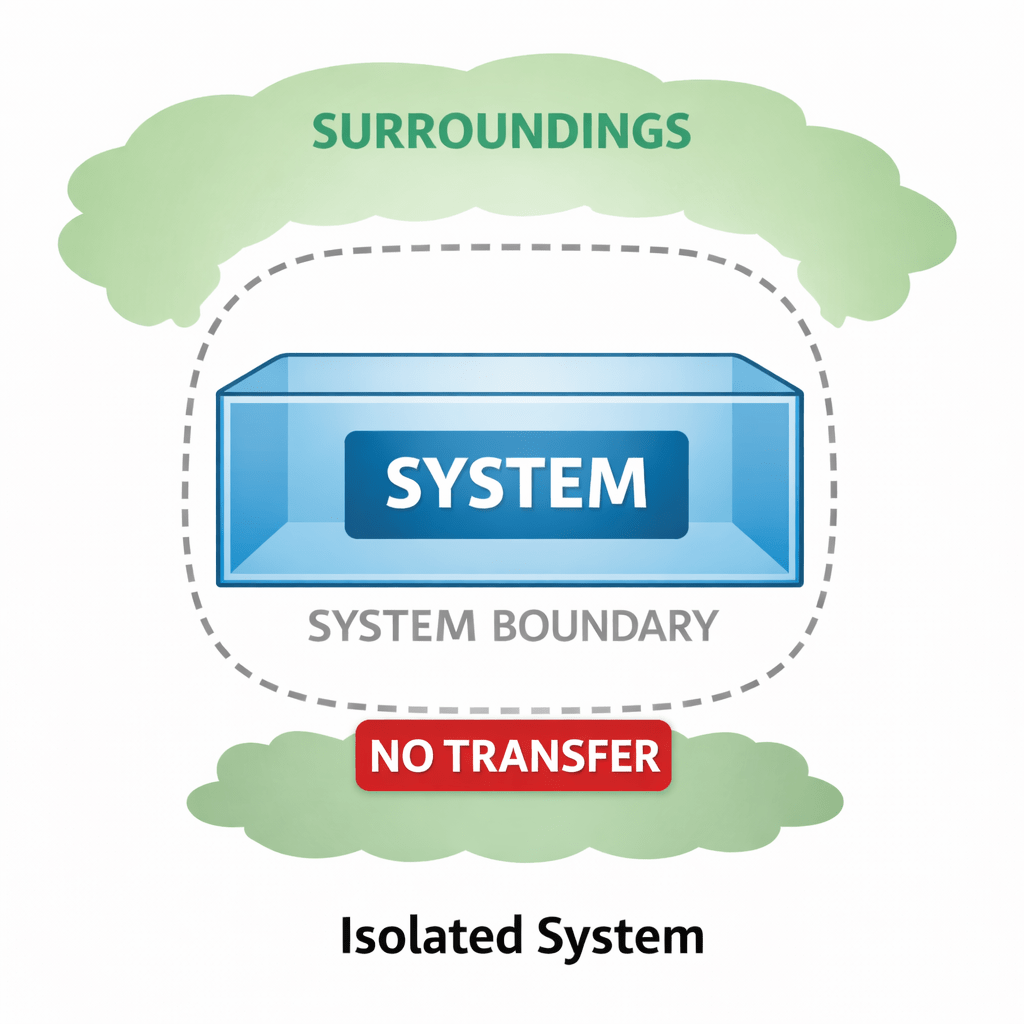
(iii) Isolated system.
The type of system in which neither matter nor energy is exchanged between system and surroundings.
Example: Substance in insulated vessel (Thermos flask)
The state of the system
In thermodynamics, the state of a system means the condition of a system at a particular moment, described by certain measurable properties.
Definition
The state of a system is the condition of the system specified by state variables such as pressure, temperature, volume, and composition.
State Variables (State Functions)
These properties determine the state of a system:
- Temperature (T)
- Pressure (P)
- Volume (V)
- Number of moles (n)
- Density
- Internal energy (U)
f these variables are known, the state of the system is completely defined.
Example
Consider a gas in a container:
- Temperature = 300 K
- Pressure = 1 atm
- Volume = 2 L
These values describe the state of the system.
Important Points
- When any state variable changes, the system goes to a new state.
- The change from one state to another is called a thermodynamic process.
Example:
Heating a gas changes temperature, pressure, or volume, so the state changes.
Short Definition for Students
State of a system:
“The set of values of thermodynamic properties such as pressure, temperature, and volume that describe the condition of a system at a given time.”
Define state function or state variables and path function.
Those properties of the system which depend only on the state of the system and not on how it is reached to that state are called state function.
Examples:
State Functions (Thermodynamic Properties)
1. Basic Thermodynamic State Variables
- Temperature (T)
- Pressure (P)
- Volume (V)
- Density (ρ)
- Number of moles (n)
- Concentration
2. Energy Functions
- Internal Energy (U)
- Enthalpy (H)
- Helmholtz Free Energy (A or F)
- Gibbs Free Energy (G)
3. Other Thermodynamic State Functions
- Entropy (S)
- Heat Capacity (C)
- Molar heat capacity
- Specific heat
Those properties of the system which depend on how the change is carried out are called path function.
Examples: :Heat energy(q),Work done(w)
Define internal energy (U).
The total energy contained by the system is called internal energy.
U=Kinetic energy + Potential energy
Internal Energy Includes
- Translational kinetic energy of molecules
- Rotational energy
- Vibrational energy
- Electronic energy
- Nuclear energy
- Intermolecular potential energy
Kinetic energy=translational energy + rotational +vibrational.
Internal energy does not include:
- Macroscopic kinetic energy
- Macroscopic potential energy
Thermodynamic Processes (JEE / NEET)
1. Isothermal Process
Temperature remains constant.
Condition:
For an ideal gas:
PV = constant
Key Points
- Heat exchange occurs.
- Internal energy change = 0 (for ideal gas).
Example
Slow expansion of gas in contact with a heat reservoir.
2. Adiabatic Process
No heat exchange between system and surroundings.
Condition:
Equation:
Key Points
- Temperature changes.
- Work done comes from internal energy.
Example
Rapid compression of gas.
3. Isobaric Process
Pressure remains constant.
Condition:P=constant
Work done:
Key Points
- Volume and temperature change.
Example
Heating gas in a cylinder with movable piston.
4. Isochoric Process (Isovolumetric)
Volume remains constant.
Condition:
Key Points
- No work done
- Heat supplied increases internal energy.
Example
Heating gas in a rigid container.
Other Processes (Sometimes Asked)
Cyclic Process
System returns to initial state.
Example
Heat engine cycle.
Reversible Process
- Infinitely slow process.
- System always in equilibrium.
Irreversible Process
- Fast process.
- System not in equilibrium.
Example
Free expansion of gas.
What is adiabatic system?
The types of the system in which there is no heat exchange between the system and surroundings through its boundary is called adiabatic system.
What is the difference between adiabatic system and isolated system.
Adiabatic system is a specified condition
Mention the sign convention for different thermodynamics function.
Energy absorbed by the system ‘q’=+ve
Energy released by the system ‘q’ =-ve
Work done by the system ‘w’ = -ve
Work done on the system ‘w’ = +Ve


Define first Law of thermodynamics.
The First Law of Thermodynamics is based on the principle of conservation of energy. It states that energy can neither be created nor destroyed; it can only be converted from one form to another. Internal energy change of the system is equal to the energy provide to the system and work done by the system.
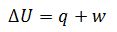
Obtain a formula for mechanical work (pressure- volume work) against constant external pressure .

What do you mean by free expansion?
Expansion of a gas in vacuum when is called free expansion.
No work during free expansion of an ideal gas whether the process is reversible or irreversible.
Define reversible and irreversible process.
The process which occur infinitely slowly such that system and surrounding are always in equilibrium with each other. The process can be reversed at any moment by an infinitesimal change called reversible process. An irreversible process can be defined as a process in which the system and the surroundings do not return to their original condition once the process is initiated

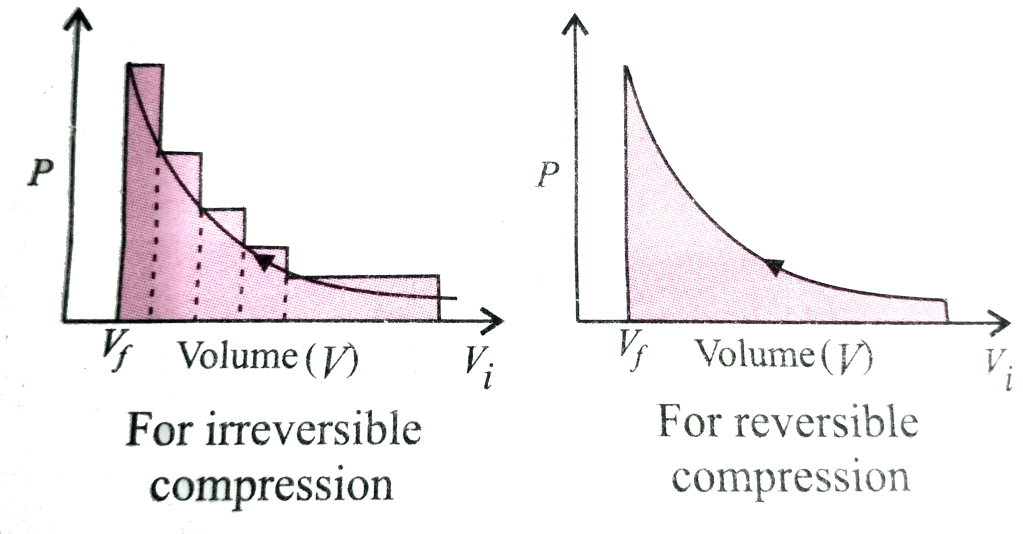
Change in internal energy of a system when
(i)Work done by the system or on the system and heat is absorbed by the system.
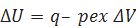
(ii)If a process is carried out at constant volume and heat is absorbed by the system.

(iii) For isothermal process.
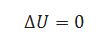
(iv) For adiabatic system.

What is the equation for calculating work done for isothermal irreversible process.
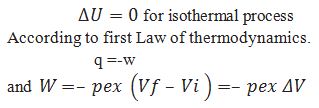
Obtain equation for the work done for the isothermal reversible process.

Define enthalpy.
A thermodynamic function, the enthalpy H is equal to H = U + pV .
U=Internal energy of the system
P=Pressure of the system.
V=Volume of the system.
What will be the change in enthalpy during the reaction at constant pressure.
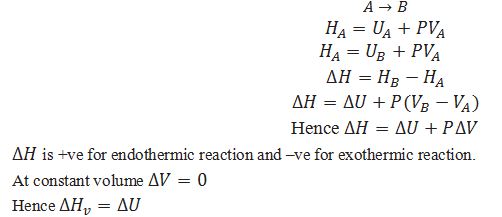
What is the equation for calculating change in enthalpy at constant pressure for a reaction involving gases.

What is extensive property and intensive properties of a system?
Those properties which depends on the amount of matter present in the system are called extensive property.Example: Mass, volume, internal energy, enthalpy.
Complete List of Common Extensive Properties
- Mass (m)
- Volume (V)
- Number of moles (n)
- Internal Energy (U)
- Enthalpy (H)
- Entropy (S)
- Gibbs Free Energy (G)
- Helmholtz Free Energy (A or F)
- Total Charge
- Heat Capacity (C)
- Total Energy
- Total Momentum
- Total Electric Charge
- Surface Area
- Length
- Weight
- Total Magnetic Moment
- Total Heat Content
Those properties which do not depend on the amount of matter present in the system are known as intensive properties.Example:Temperature, density, pressure etc.
Complete List of Common Intensive Properties
- Temperature (T)
- Pressure (P)
- Density (ρ)
- Concentration
- Refractive Index
- Boiling Point
- Melting Point
- Specific Heat Capacity
- Molar Heat Capacity
- Molar Volume
- Viscosity
- Surface Tension
- Thermal Conductivity
- Electrical Conductivity
- Specific Gravity
- Color
- Odor
- Hardness
- Molar Entropy
- Molar Enthalpy
- Molar Gibbs Energy
Define heat capacity.
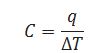
Heat requires by the substance to increase the temperature by one 0C is called heat capacity.
Define specific heat capacity.

Heat requires by the unit mass of the substance to increase the temperature by one 0C is called specific heat capacity.
Molar heat capacity is the amount of heat required to raise the temperature of 1 mole of a substance by 1 Kelvin (or 1°C).
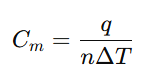
What is the relation between heat capacity at constant volume (Cv) and heat capacity at constant pressure Cp)?
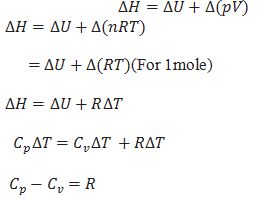
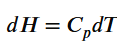
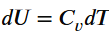
What is bomb calorimeter?
A calorimeter in which the chemical reaction is carried out at constant volume. Hence there is no work done.
How can we calculate the enthalpy change for reaction?
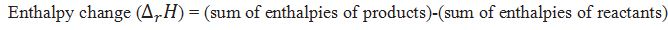
Define standard enthalpy of reaction.
The standard enthalpy of reaction is the enthalpy change that occurs on a system when one mole of matter is transformed by a chemical reaction under standard condition.
Define standard enthalpy of fusion

Enthalpy of fusion is the change in enthalpy when one mole of solid is melted in liquid.
Define standard enthalpy vaporization

Amount of heat required to vaporize one mole of liquid at constant temperature.
Define standard enthalpy of sublimation
The change in enthalpy when one mole of a solid substance is converted into vapour state at constant temperature.
Define standard enthalpy of formation
Enthalpy change during the formation of one mole of a compound from its constituent element in their standard state.

If an element is already in its most stable form at 1 bar pressure, its enthalpy of formation is defined as zero.
This is just a reference point in thermodynamics.
Examples
because these are elements in their standard states.
Define enthalpy of combustion
Enthalpy change during combustion of one mole of a substance when all the reactants and products are in standard state.
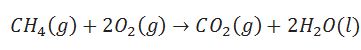
Define enthalpy of atomisation.
(i )Enthalpy change during breaking of one mole of bond of a compound completely into gaseous state.
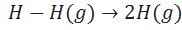
(ii)Enthalpy of atomization is also called enthalpy of sublimation in case of solid element.

Define bond dissociation enthalpy.
The bond dissociation enthalpy is the change in enthalpy when one mole of covalent bond is broken to form product in gaseous state.
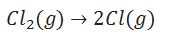
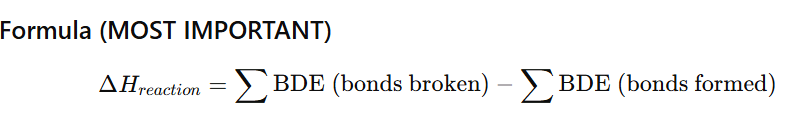
Define mean bond enthalpy.
For the polyatomic molecules mean bond enthalpy is defined as total bond dissociation enthalpies divided by number of bonds.
Define Hess’s Law of constant heat summation.
When a reaction takes place in several steps then its standard enthalpy is the sum of the standard enthalpies of the intermediate reactions.
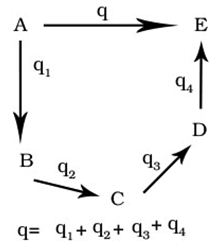
Define enthalpy of solution
Enthalpy change when one mole of substance is dissolved in given amount of solvent at give temperature.
Define lattice enthalpy.
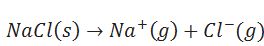
The enthalpy change when one mole of an ionic compound dissociates into it constituent ions in gaseous state.
Define enthalpy of hydration.
The energy released when new interacting are made between the gaseous ions and water molecules is called hydration enthalpy.
What is the relationship between enthalpy of solution,lattice enthalpy,hydration enthalpy.

For ionic compound
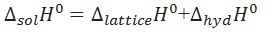
Criteria for solubility of ionic compounds in water
Main Solubility Criterion
An ionic compound dissolves in water when:
Meaning:
Hydration energy ≥ lattice energy
Then dissolution becomes energetically favorable.
What is Born -Haber cycle.
With the help of Born-Haber Cycle we determine the lattice enthalpy of the substance.

Heat of Neutralisation
1. Definition
Heat of neutralisation is the enthalpy change when 1 mole of water is formed from the reaction of an acid and a base.
2. Standard Value (Most Important Point)
3. Strong vs Weak (Very Important)
Case 1: Strong acid + Strong base
Example: HCl + NaOHΔH=−57.1kJ/mol
Case 2: Weak acid + Strong base
Example: CH₃COOH + NaOH
Case 3: Strong acid + Weak base
Example: HCl + NH₄OH
Same logic → less heat released.
Case 4: Weak + Weak
Least heat evolved.
4. Key Formula (Numerical)
5. Limiting Reagent Concept (Very Important)
Heat depends on moles of water formed, not volume.
Example:
- If H+ = 0.1 mol
- OH− = 0.05 mol
→ Water formed = 0.05 mol
6. Temperature Rise Formula
Trap 1
Students think more volume = more heat ❌
✔ Depends on moles reacting
Trap 2
Using -57.1 for weak acids ❌
✔ Only for strong + strong
Trap 3
Forgetting diprotic acids
Example:
Define spontaneous reaction.
The reaction which occurs itself without any external force is called spontaneous reaction.
Examples:Burning of carbon in oxygen.Cooling of hot water.
What do you mean by spontaneity of chemical reaction?
Spontaneity means feasibility of particular chemical reaction that is that reaction is possible or not.
What is the criteria for spontaneous process?

Define entropy(s)
The degree of randomness or disorderness is called entropy.
How can we calculate the change in entropy of a reversible reaction?
Where q=enthalpy change during chemical reaction or heat released or absorbed by the system.
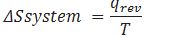
What is the relation between change in entropy of the system ,change in entropy of surrounding )?
What is the relation between change in entropy of the system. change in enthalpy of system ) and Gibbs energy change ?
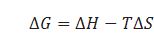
In that case in which randomness increases the change in entropy is taken as +ve and when randomness decreases the change in entropy is taken as –ve.
What is the formula for calculating equilibrium constant Kc?
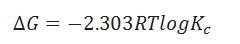
When the particular reaction is non spontaneous, spontaneous and equilibrium according to .
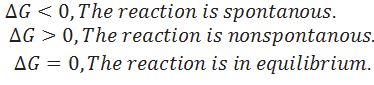
Predict whether the reaction may occur spontaneously or not in following cases.

ALL FORMULAS OF ENTROPY CHANGE (ΔS)


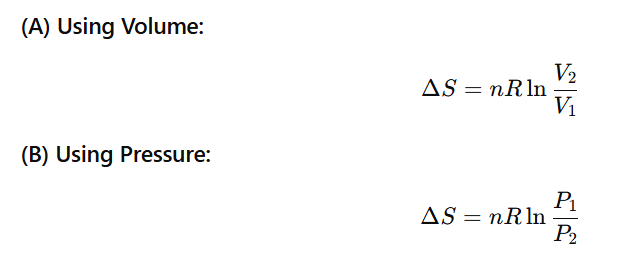

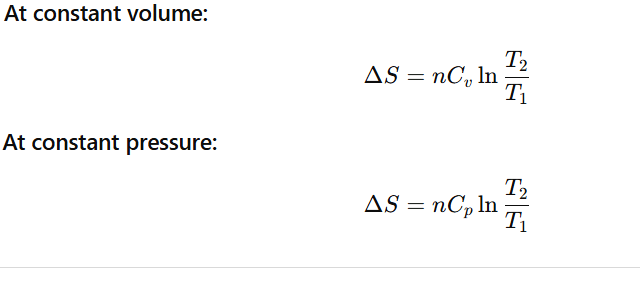
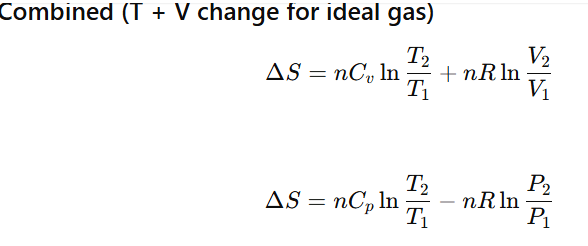
All important formulas for an adiabatic process (very useful for JEE/NEET)
1. Basic Condition (Definition)
- No heat exchange
q=0
From First Law of Thermodynamics:ΔU=−W
2. Pressure–Volume Relation (Ideal Gas)
- (adiabatic index)
3. Temperature–Volume Relation
4. Temperature–Pressure Relation
Work Done in Adiabatic Process
✔ General formula:
✔ In terms of temperature:
6. Change in Internal Energy
7. Relation between P, V, T (Combined)
8. For Reversible Adiabatic Process
- Also called Isentropic process
9. Important Points (Exam Tricks)
- Temperature decreases in expansion
- Temperature increases in compression
- Adiabatic curve is steeper than isothermal
- No heat transfer → fast process
